

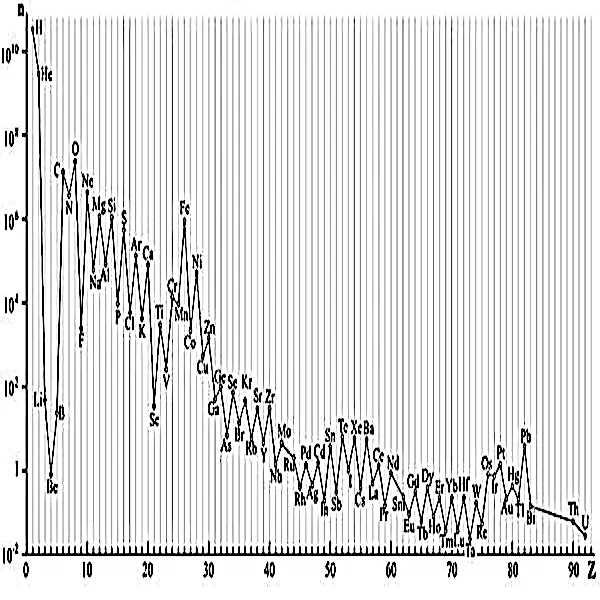
In the first moments after the Big Bang, about 13.8 billion years ago, the Universe was a soup of quarks and gluons of unimaginable density and temperature. As it cooled, the first protons and neutrons assembled. During the first three minutes, the cosmos was the stage for a rapid primordial nucleosynthesis. The result of this original alchemy is absolutely simple: about 75% hydrogen (¹H) and 25% helium (⁴He), with tiny traces of lithium and beryllium.
This initial legacy, this primordial abundance, is the basic score on which the entire cosmic history will be written. No element heavier than lithium could emerge from this era. To create carbon, oxygen, iron, and all the atoms that now make up planets, oceans, and our own bodies, we had to wait for the birth, life, and violent death of the first stars. Margaret Burbidge (1919-2020), a pioneering astrophysicist, profoundly influenced the study of the formation of chemical elements within stars and the structure of galaxies. Thanks to her research, we now know that we are, literally, made of "stardust".
| Element | Symbol | Relative abundance (atoms) (normalized to 10⁶ silicon atoms) | Mass ratio (fraction in %) | Main production sites |
|---|---|---|---|---|
| Hydrogen | H | \( 4.0 \times 10^{10} \) | ~ 71 % | Big Bang (primordial nucleosynthesis) |
| Helium | He | \( 3.0 \times 10^{9} \) | ~ 27 % | Big Bang, stellar fusion |
| Oxygen | O | \( 2.2 \times 10^{7} \) | ~ 0.9 % | Massive stars (C, Ne, O fusion), supernovae |
| Carbon | C | \( 1.0 \times 10^{7} \) | ~ 0.3 % | Low-mass stars (triple alpha process), massive stars |
| Neon | Ne | \( 1.8 \times 10^{6} \) | ~ 0.1 % | Massive stars (carbon fusion) |
| Nitrogen | N | \( 6.2 \times 10^{5} \) | ~ 0.09 % | Low-mass stars (CNO process), massive stars |
| Silicon | Si | \( 1.0 \times 10^{6} \) | ~ 0.07 % | Massive stars (oxygen fusion, silicon fusion) |
| Magnesium | Mg | \( 1.0 \times 10^{6} \) | ~ 0.06 % | Massive stars (neon and carbon fusion) |
| Iron | Fe | \( 8.3 \times 10^{5} \) | ~ 0.12 % | Supernovae (core collapse), type Ia supernovae |
| Sulfur | S | \( 4.8 \times 10^{5} \) | ~ 0.04 % | Massive stars (oxygen and silicon fusion) |
| Argon | Ar | \( 1.0 \times 10^{5} \) | ~ 0.01 % | Massive stars (silicon fusion) |
| Nickel | Ni | \( 5.0 \times 10^{4} \) | ~ 0.008 % | Supernovae (core collapse, type Ia) |
| Calcium | Ca | \( 5.0 \times 10^{4} \) | ~ 0.005 % | Massive stars (silicon fusion) |
| Aluminum | Al | \( 4.5 \times 10^{4} \) | ~ 0.003 % | Massive stars, supernovae |
| Sodium | Na | \( 2.2 \times 10^{4} \) | ~ 0.001 % | Massive stars, AGB stars |
| All other elements | — | < \( 1.0 \times 10^{4} \) | < 0.002 % | Supernovae, white dwarf mergers, neutron star collisions, s-process |
N.B.: Abundance values are based on spectroscopic data from the Sun and the interstellar medium, which is the standard reference in astrophysics. Abundance is normalized to \( 10^6 \) silicon (Si) atoms, a common convention in astronomy. Hydrogen and helium alone account for about 98% of the ordinary baryonic mass of the Universe. The mass fraction of elements heavier than helium ("metallicity") is therefore about 2% in the solar neighborhood.
Stars are the alchemical reactors of the Universe. In their incandescent cores, pressure and temperature are so high that atomic nuclei fuse. This reaction, governed by the balance between gravity and radiation pressure, follows a precise hierarchy.
Hydrogen fuses into helium, releasing the energy that makes stars shine. When the core hydrogen is exhausted, a massive star (more than 8 solar masses) begins to fuse helium into carbon, then carbon into neon, and so on, forming concentric layers like an onion. This fusion chain stops at iron \( (^{56}\text{Fe}) \), the most stable element. Iron is the nuclear graveyard: its fusion no longer releases energy, it consumes it. The iron core then collapses abruptly, triggering a massive supernova explosion (type II).
It is in this fraction of a second and in the stellar winds that follow that all elements heavier than iron are forged. The periodic table, from cobalt to uranium, is the fruit of these stellar cataclysms. Without supernovae, the Universe would have remained a sterile place, composed only of hydrogen, helium, and a few traces of lithium.
| Nucleosynthesis site | Main elements produced | Physical process | Role in cosmic evolution |
|---|---|---|---|
| Big Bang (early moments) | Hydrogen (¹H), Helium (⁴He), traces of Lithium (⁷Li) | Primordial nucleosynthesis | Foundation of all baryonic matter, initial composition of the cosmos. |
| Low-mass stars (< 8 M☉) | Helium (⁴He), Carbon (¹²C), Nitrogen (¹⁴N) | Hydrogen and helium fusion, s-process (slow) during the AGB phase | Enrichment of the interstellar medium in light elements, essential for organic chemistry. |
| Massive stars (> 8 M☉) | Oxygen (¹⁶O), Silicon (²⁸Si), Magnesium (²⁴Mg), up to Iron (⁵⁶Fe) | Hydrogen, helium, carbon, neon, oxygen, silicon fusion | Creation of the elements that make up rocks and planetary cores. |
| Supernovae (core collapse) | Elements heavier than iron: Nickel (⁵⁸Ni), Cobalt (⁵⁹Co), Zinc (⁶⁴Zn), up to Uranium (²³⁸U) | Rapid neutron capture (r-process) and explosive shock wave | Dispersion of heavy elements in the interstellar medium, essential for technologies and radioactivity. |
| White dwarf mergers (type Ia) | Iron (⁵⁶Fe), Nickel (⁵⁸Ni), iron group elements | Explosive combustion of carbon and oxygen | Major contribution to iron abundance, chemical marker of the age of stellar populations. |
The abundance of chemical elements is not just a catalog. It is a tool for stellar archaeology. By analyzing the light from a star, its spectrum, astrophysicists can determine its chemical composition. This chemical signature acts as a fossil, revealing the time of its birth.
The oldest stars, formed more than 10 billion years ago, are extremely poor in metals (elements heavier than helium). These are Population II stars, often found in the galactic halo and globular clusters. In contrast, young stars, like our Sun, are rich in metals (Population I), testifying to successive cycles of nucleosynthesis and enrichment.
A fascinating discovery is that of "ultra-metal-poor" stars, such as SMSS J031300.36-670839.3. Discovered in 2014, it has an iron abundance more than a million times lower than that of the Sun, but a relatively high amount of carbon. This suggests that its raw material came from a first generation of massive stars (Population III) that enriched the medium with carbon before exploding as supernovae, but without producing much iron. This is direct evidence of the diversity of physical processes at work in the early Universe.
Galactic collisions and mergers also play a major role. When two spiral galaxies collide, as in the case of Arp 220, huge clouds of gas are compressed, triggering bursts of massive star formation. These stars, which live fast and die as supernovae, suddenly enrich the medium with heavy elements. Chemical abundance is therefore not static: it is the dynamic reflection of the gravitational and evolutionary history of each galaxy.
The emergence of life, as we know it, is a direct consequence of this long chemical evolution. Carbon, the central element of organic chemistry, is the product of helium fusion in stars (triple alpha process). Oxygen, which we breathe, is the third most abundant element in the Universe, forged in the cores of massive stars. Nitrogen, a component of amino acids, is synthesized during CNO (carbon-nitrogen-oxygen) cycles in stars.
The formation of rocky planets is itself conditioned by the abundance of refractory elements such as silicon, iron, magnesium, and aluminum. In regions of the Universe where metallicity is too low, the probability of forming terrestrial planets collapses. We can therefore trace a direct link, a physical causality, between the rate of supernova formation, the chemical enrichment of the interstellar medium, and the possibility of the emergence of habitable worlds. Carl Sagan's famous phrase, from his book Cosmos (1980), finds its deepest root here: "We are all made of star stuff."